‘Drug’-induced interstitial lung disease in a patient with a heterozygote P450 polymorphism
Khaled Mansour1, Petal Wijnen2, Kitty Linssen3, Aalt Bast4, Marjolein Drent1. Dept of Respiratory Medicine and ild care team1, Dept of Clinical Chemistry2, Dept of Medical Microbiology3, Dept of Pharmacology and Toxicology4, University Hospital of Maastricht, Maastricht, the Netherlands.
Introduction
The lung is a major target for inhaled agents and toxicants. Many inhaled chemicals are not hazardous as such, but are biotransformed to reactive intermediates [1]. Acute and chronic parenchymal lung injuries as a result of the inhalation of illicit drugs are not uncommon complications in habitual drug users. A variety of interstitial and other long diseases may occur in such cases with diverse clinical presentations.
Case history
A 22-years old male presented with dyspnea, non-productive cough, myalgia and fever. These symptoms started 4 days prior to admission. His medical history was unremarkable. He smoked tobacco for a few years. Finally, he admitted alcohol abuse, heavy marijuana and cocaine smoking in the last months. Physical examination revealed some crackles at auscultation, he was a little tachypnoeic (20 breaths•min–1) and his body temperature was 36.8°C. He had a moderate hypoxemia (pO2 8.7kPa, breathing room air), high serum C-reactive protein (254 mg/l), a white blood cell count of 16.3 109•L–1 and a normal number of eosinophils. Figure 1 shows his chest roentgenogram demonstrating diffuse infiltrates at presentation. Figure 2 shows his high resolution CT (HRCT) scan with a diffuse reticulonodular pattern ad admission. Spirometry two days after admission showed normal dynamic lung volumes with slightly decreased diffusing capacity (81% of predicted).
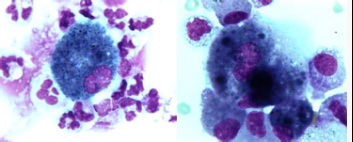
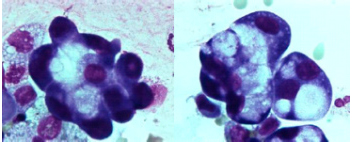
A bronchoscopy showed no endobronchial abnormalities. Bronchoalveolar lavage fluid (BALF) cell differentiation revealed a high number of polymorphonuclear neutrophils (PMNs; 32.4% of the total cell count with 53.8% alveolar macrophages (AMs), 11.2% lymphocytes (Lyms), 2.0% eosinophils (Eos) and 0.2% mast cells (MC). Moreover, some ‘foamy’ AMs and a few reactive pneumocytes type II (PII cells) were seen (figure 3). No intracellular micro-organisms were seen. Moreover, cultures remained sterile. Serology for HIV infection was negative, as well as the urinary test for Legionella pneumonia and Streptococcus pneumoniae. Serology for common virus, and mycoplasma, ricketsiae and chlamydiae was negative. No underlying immunosuppressive condition was evident.
Additionally, relevant CYP450 polymorphisms were profiled. This revealed that he appeared to be a CYP2C19 intermediate metabolizer (*1/*2). The clinical consequences obtained from the software package www.genemedrx.com are summarized in table 1.
Table 1. The ‘drug’-drug and ‘drug’-gene interactions of the presented case, a CYP2C19 intermediate metabolizer (*1/*2)
| Affected drugs | Change | Causative agent |
| cocaine | > 150% ? | resveratrol* |
| marijuana | 0% | |
| azitromycin (Zithromax) | > 150% ? | resveratrol |
| ibuprofen | 0-25% | marijuana, resveratrol |
*resveratrol: red wine component
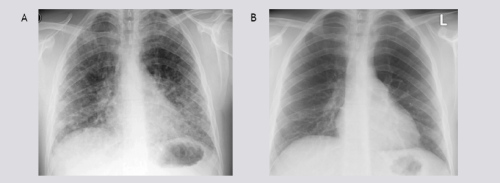
Figure 1 A) Chest radiograph at admission shows parenchymal lung opacities; B) Two weeks after admission, normalized.
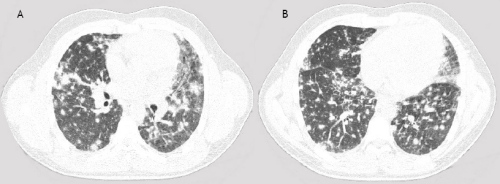
Figure 2 The HRCT shows irregular nodular abnormalities, no signs of pleural effusion or hilar node enlargement.
Treatment and clinical course
The patient was initially treated with a broad-spectrum antibiotic, moxifloxacine intravenously, aiming to cover the majority of common and atypical microbes. Because of high suspicion of a ‘drug’-induced interstitial lung disease (DI-ILD) and the absence of features of any infectious cause, corticosteroids were started (40mg daily, i.v.) and continued for two weeks. Thereafter, the corticosteroids were tapered gradually. His clinical condition improved within 2 weeks. Dyspnea and cough disappeared, follow-up chest radiograph abnormalities cleared (figure 1B) and the PaO2 became within normal limits (11.9 kPa; on room air at rest).
|
Figure 3A |
Figure 3B |
Figure 3 A) May-Grünwald Giemsa stained cytocentrifuged; B) Reactive type II pneumocyte (PII cell). BALF sample showing alveolar macrophages (AMs) containing phagocytised material. Magnification 1000x.
Discussion
A variety of interstitial lung damage due to cocaine or marijuana inhalation, ranging from acute pulmonary haemorrhage to chronic eosinophilic infiltrates have been described [1,2].
To date, the mechanism of lung injury is not fully understood and a direct toxic effect in a dose-dependent pattern of the inhaled agent is the most suspected [3].
Several different xenobiotic-metabolizing CYP450 and phase II enzymes (i.e. conjugation enzymes including several transferases) are present in the human lung and lung-derived cell lines, possibly contributing to in situ activation and
inactivation of toxicants. CYP2C19 belongs to the largest CYP450 family and together with other members like CYP2D6, and CYP2C9 metabolizes, to varying amounts, more than half of all frequently prescribed drugs [4]. It has also a significant role in the detoxification of many xenobiotica such as alcohol [5] or marijuana and its metabolites [6].
This patient was tested as having a heterozygote CYP2C19 variant allele. This means that he is an intermediate metabolizer for marijuana and cocaine. Simultaneously with marijuana and cocaine, he used excessive alcohol for several months.
It is tempting to speculate that the development of interstitial damage after exposure to toxic agents in this case, was associated with being an intermediate metabolizer of CYP2C19.
Conclusions
This case report highlights the increasing importance of introducing pharmacogenomics into the work-up of patients with suspected drug-induced toxicity.
Interindividual differences in the expression of CYP450 enzymes may contribute to the risk of developing interstitial and other lung diseases initiated by agents that require metabolic activation.
CYP450 polymorphism should be considered as one of the pathways in the development of adverse drug reactions and sometimes toxic effects.
References
Tashkin DP. Airway effects of marijuana, cocaine, and other inhaled illicit agents. Curr Opin Pulm Med 2001; 7: 43-61.
Gotway MB, Marder SR, Hanks DK, Leung JW, Dawn SK, Gean AD, Reddy GP, Araoz PA, Webb WR. Thoracic complications of illicit drug use: an organ system approach. Radiographics; 2002:S119-35.
Tashkin DK. Smoked marijuana as a cause of lung injury. Mondali Arch Chest Dis 2005; 63: 93-100.
Nebert DW, Russell DW. Clinical importance of the cytochrome P450. Lancet 2002; 360:1155-62.
Hamitouche S, Poupon J, Dreano Y, Amet Y, Lucas D. Ethanol oxidation into acetaldehyde by 16 recombinant human cytochrome P450 isoforms: role of CYP2C isoforms in human liver microsomes. Toxicol Lett 2006: 167:221-30.
Bland TM, Haining RL, Tracy TS, Callery PS. CYP2C-catalyzed delta9-tetrahydrocannabinol metabolism: kinetics, pharmacogenetics and interaction with phenytoin. Biochem pharmacol 2005; 70: 1096-103.
Source: Mansour E, Wijnen P, Linssen K, Bast A, Drent M. ‘Drug’-induced interstitial lung disease in a patient with a heterozygote P450 polymorphism. 16-09-2007. Thematic poster
session: ‘Considerations in the management of IPF and other ild’. Eur Respir J 2007; 30:118s.
Feedback & comments
If you would like to share any comments on this case please send them to wasog@gosker.nl and after peer review your comment(s) will be posted below.